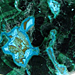
Photo of Fluorite in Processed & Rough Form
Photo of Fluorite in Processed & Rough Form
Fluorite is a mineral with a hardness of 4 out of 10 on the Mohs scale of mineral hardness [?]. These Cubicly structured gems are made of calcium floride, their full chemical compound being CaF2.
Fluorite is a widely-distributed mineral in the Halide classification, and is now mined in vast quantities. It is a suitable mineral for collectors and is rarely cut as a gem, but massive varieties are carved as ornamental objects.
It appears in cubes, octahedrons, dodecahedrons, while other forms are rarer. Compact, banded, and concretionary masses are frequently found. Most fine crystals of fluorite are not faceted (though faceted stones can be very beautiful) as fine groups sell for high prices.
Its color is variable, colorless when pure, tinted by impurities to blue, pink, purple, green, or yellow. It has a vitreous luster, and parallel to octahedron cleavage.
It is semi-hard, fragile, has perfect octahedral cleavage, which produces triangular shapes on the corners of the cubic crystals when broken. It is transparent to translucent with vitreous luster.
It is insoluble in water, hydrochloric acid, and nitric acid, but is soluble in sulphuric acid, giving off fumes of hydrofluoric acid, often strongly fluorescent under ultraviolet light, when heated to temperatures above 100 C. It fuses easily, at least around the edges, turning the flame brick red.
Fluorite is found in sulphide deposits as gangue with barytes and quartz. It occurs in medium- and high-temperature hydrothermal veins associated with lead and zinc. It is rarer in pegmatites, where it is associated with cassiterite, topaz, tourmaline and apatite. It occurs as an accessory mineral in cavities of many felsic and intermediate intrusives, and as a sublimation product of volcanic rocks. It may also have sedimentary origin, probably by deposition in enclosed basins from saline waters possibly of volcanic origin. Fluorite crystals are included in topaz and opals.
Splendid crystals are found in various localities. Colorless cleavage fragments of optical quality from Corvara in Val Sarentina (Bolzano, Italy); pink octahedrons from the St. Gotthard (Switzerland); green octahedrons from Cera (Brazil), Kongsberg (Norway) and Bancroft (Canada); green, white, or violet cubes from Cumberland, Derbyshire (England); yellow cubes from Wolsenberg (Germany); purple crystals from Madoc (Canada) and various places in the Alps, especially near deposits of radioactive minerals (e.g. Valle Imagna, Bergamo, Italy).
Significant masses worth mining are found in Ontario (Canada), Illinois and Kentucky (USA), England, Soviet Republics, Mexico, and Italy.
The best known type of fluorite is Blue John (from Derbyshire,England), also a violet-blue and white or yellowish banded variety used in jewellery and vases as far as Roman times. It is also known as "derbystone," "Derbyshire spar," "cand," "cann."
Fluorite is also known as fluorspar (its old name), and is confused with many gemstones. Other names for fluorite include "cabra stone,"
Misnomers for fluorite include "Beryllfluss," "Bohemian emerald," "Bohemian topaz," "African emerald," "emeraude morillon," "esmeralda de Cartagena," "false amethyst," "false emerald," "false ruby," and many others.
The specific gravity [?] for Fluorite is 3.18, its refractive index [?] is 1.43, and its double refraction [?] is None.
History
Its name comes from the Latin "fluere" meaning "to flow," which refers to its use as a flux. Miners once called the beautifully colored crystals "ore flowers". As the name suggests, it can be strongly fluorescent in ultraviolet light.
The Benedictine Abbey Emerald is an emerald green fluorite weighing 12.90 kg given to Benedictine Abbey.
Industrial Usages
Fluorite is an important raw material used to prepare several fluorine compounds, such as hydrofluoric acid. It is also used in the manufacture of pottery, in the optical and plastics industries, and in the metallurgical treatment of bauxite. Lower-grade fluorite is used as a flux in steelmaking and foundry work.
Virtually all fluorite octahedra found on sale have been cleaved by dealers.
Very clear crystals are used as apochromatic lenses and as spectographic prisms.
There are large reserves of fluorite of potential industrial use in dried-up lake basins in southern Tuscany and Lazio (Italy).
Synthetic fluorites of very high purity can irradiated with energetic radiation to produce desirable colorations.